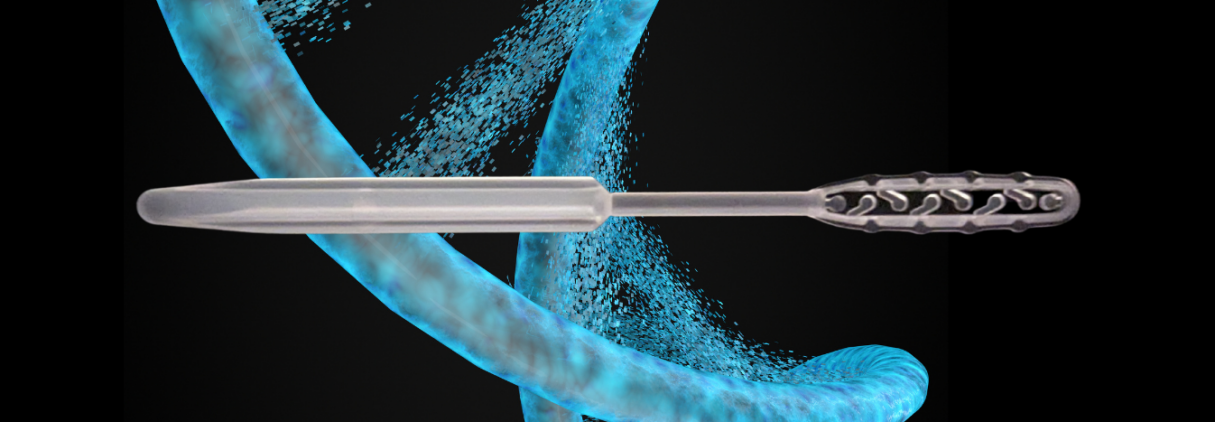
“The full release capacity functionality allows for adequate sample capture needed for DNA sample collection and processing.”
Mawi DNA Technologies, a medical device company focusing on the development of innovative technologies for biosampling, has launched the BioWAND, a 100% medical-grade plastic swab designed for high-efficiency capture and release of oral DNA samples without the possibility of contamination from the glue or fibers used in flocked and cotton swabs.
The COVID-19 pandemic created major supply chain disruptions that are still relevant today, especially for swabs. In response to these supply issues, Mawi DNA Technologies developed the 100% plastic, injection-molded BioWAND as an efficient alternative to standard oral swabs.
“The BioWAND sterile sampling applicator is three inches in length with a unique head design positioned at the distal end and an integrated breaking point,” said Dr. Bassam El-Fahmawi, CEO of Mawi “The swab head is designed with a channel separated with teeth that retains the collected oral sample which can then be released into the transport media with a gentle rotation. The full release capacity functionality allows for adequate sample capture needed for DNA sample collection and processing downstream from manual and robotic liquid handling systems.” Dr. El-Fahmawi added.
To support the high demand for medical-grade sample collection swabs, the BioWAND’s supply chain line remains reliable and stable due to the plastic molded construction and USA-based manufacturing.
Helix Partners with Mawi DNA Technologies to Expand Access to…...
SAN MATEO, Calif. and PLEASANTON, Calif., Feb. 10, 2026 -- READ MORE
Safer DNA Sample Stabilization Starts with Mawi...
A Journey of Innovation and Vision – The Mawi Story…...
Mawi was founded with one major goal: to simplify sample collection, transportation, and storage while making the process as painless as possible, especially for babies and the elderly. The collected ...
READ MORE